
The kinetic theory of gases, in contrast to Dalton's atomic theory, defines how gases behave physically, such as through diffusion, viscosity, conductivity, pressure, etc., rather than how they interact chemically to produce compounds. Assuming that these were the basic building blocks of matter, he chose to call these units "atoms." Early in the 19th century, John Dalton, a chemist, found that chemical elements appeared to combine with one another by basic units of weight. These antiquated ideas are not the foundation of modern atomic physics. Rather than being founded on empirical evidence, this ancient concept was based on philosophical reasoning. It is known that the word "atom" originated from the Greek word "atomos." Atomos means uncuttable, defined because an atom is the smallest particle and cannot be made smaller. For example, a tennis ball's behavior is uncertain to predict. Using classical physics, it is almost impossible to detect the exact behavior of an atom due to its small size, which causes quantum effects. The size of an atom is very small, approximately 100 picometers. The only element free of a neutron is hydrogen.Ītom forms every object, whether solid, liquid, or gas. The other components present in an atom are protons and neutrons, which are present inside the nucleus. 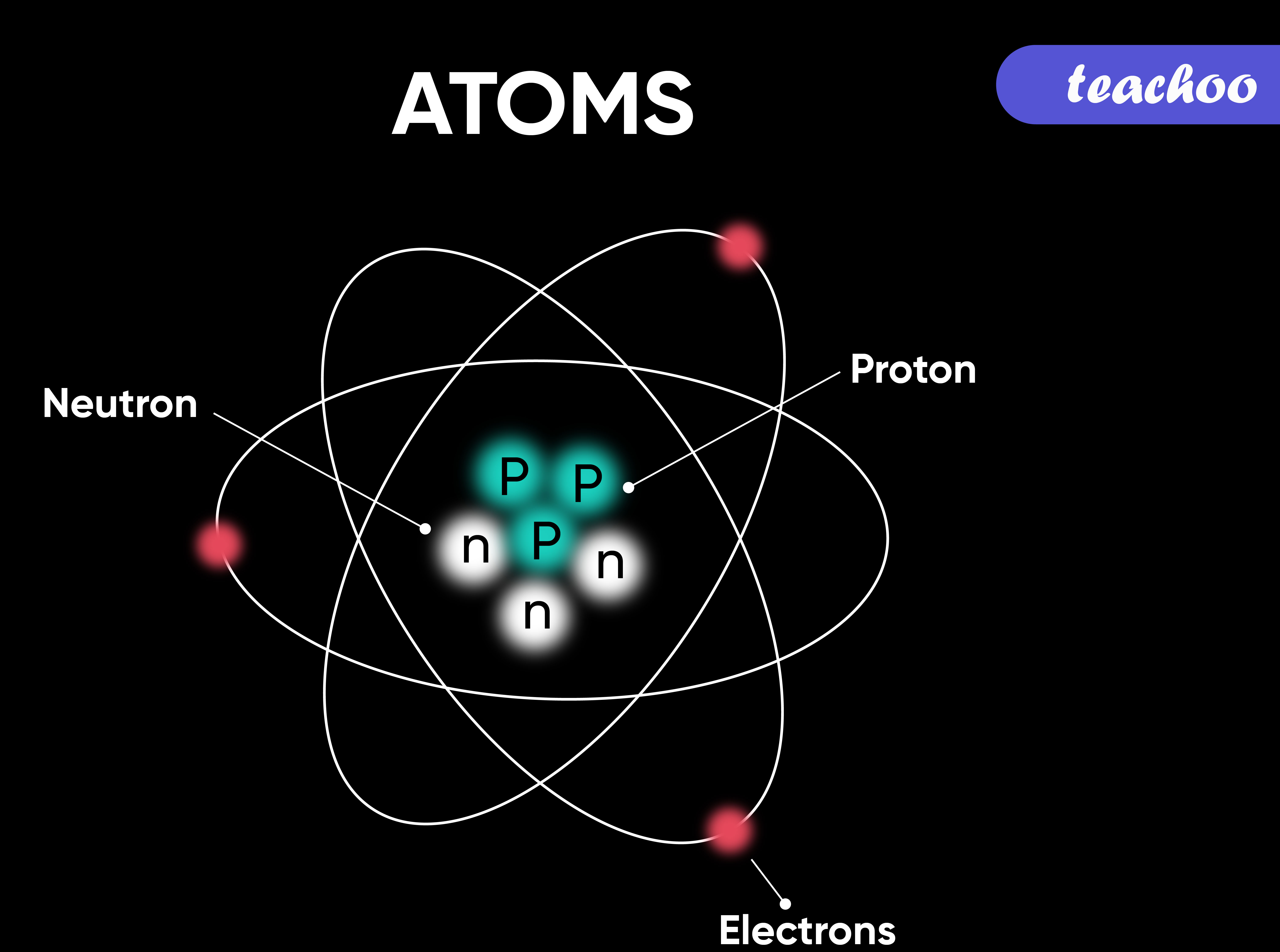
Each atom has electrons associated with the nucleus. Atom represents the fundamental unit of any matter existing in this universe. All the chemical elements in this world are made of a universal unit called an atom.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
